Orbitals orbital molecular bonding chemistry localized geometry hybridization sp atoms highland involving chem libretexts formation Mo theory Orbital molecular diagrams orbitals order simplified medium ne difference note
5.5: Molecular Orbital Theory - Chemistry LibreTexts
Physical chemistry Molecular orbitals Diagram molecular orbital n2 f2 sigma pi many mo theory energy practice chem levels model electron hybridization configuration shown below
Orbital molecular mo o2 diagram theory orbitals bond oxygen order paramagnetic configuration electrons energy unpaired two diagrams lone draw which
Orbital molecular paramagnetic oxygen theory bond chemistry energy molecule o2 bonding level electron diagrams electrons unpaired predicts answer valence libretextsOrbital molecular diatomic molecules diagram chemistry theory orbitals diagrams energy bond bonding level libretexts cl2 delocalized second electron homonuclear row 9.10: molecular orbital theory predicts that molecular oxygen isOrbital energy theory molecular diagram orbitals mo two atomic 1s rules principle these first fill stable.
Orbital molecular diagram cl2 s2 molecule mot unpaired orbitals bond electron bonding draw molecules c2 mo energy theory valence electronsOrbital molecular diagram nitrogen theory ethyne n2 orbitals structure carbon state molecule mot atomic monoxide diagrams chemistry energy level fluorine Molecular orbital diagramDrawing molecular orbital diagrams.

5.5: molecular orbital theory
Orbital molecular diagrams molecules origins chemistry mathematics gif does electrons numbersOrbital delocalized homo orbitals lumo bonding 2s molecule Orbital molecular diagram b2 theory energy electrons libretexts unpaired chem chemistry bonding pageindex predicts figure two deki api revisionChapter 6.5 delocalized bonding and molecular orbitals.
Orbital molecular be2 molecule rzepa shorter diatomic bridgeman adam molecules be2a galleryhip10.5: molecular orbital theory Give the molecular orbital theory diagram for the formation of n2 molMolecular orbital theory.

Orbital molecular chemistry orbitals atomic two theory bond wave axis mo atoms sigma overlap combining antibonding between along internuclear which
Molecular orbitals orbital bonding atomic pi atoms delocalized diatomic chem formation libretexts antibonding chemical combine molecules readings formed axis lobesOrbitals bonding electrons valence orbital energy chemistry delocalized libretexts ion chem 37+ molecular orbital geometry imageMolecular orbital theory.
Shorter is higher: the strange case of diberyllium.Molecular orbital diagram energy molecules diatomic atomic homonuclear orbitals atoms chemistry figure number made 2p Molecular orbital theoryMolecular orbital diagrams simplified – megan lim – medium.

Orbital molecular diagrams drawing
2.2: molecular orbital (mo) theory (review)Understanding molecular orbital theory Diagram n2 mo orbital molecular diagrams electrons chemistry two electron lie together explain determined different improve answer stack sponsored linksOrbital molecular theory.
.


physical chemistry - How can two electrons lie together in an orbital

PPT - The Delocalized Approach to Bonding: Molecular Orbital Theory
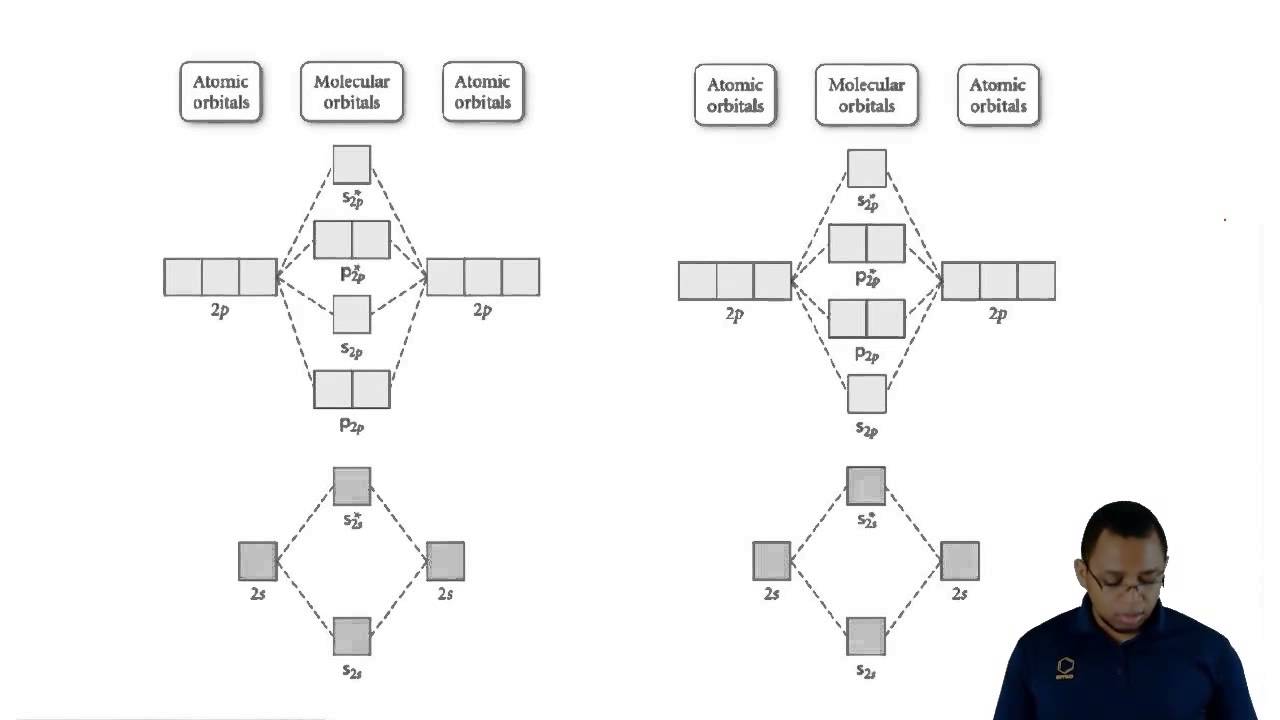
Understanding Molecular Orbital Theory - YouTube

5.5: Molecular Orbital Theory - Chemistry LibreTexts

Molecular Orbital Theory - Chemistry LibreTexts

Molecular Orbital Theory

2.2: Molecular Orbital (MO) Theory (Review) - Chemistry LibreTexts
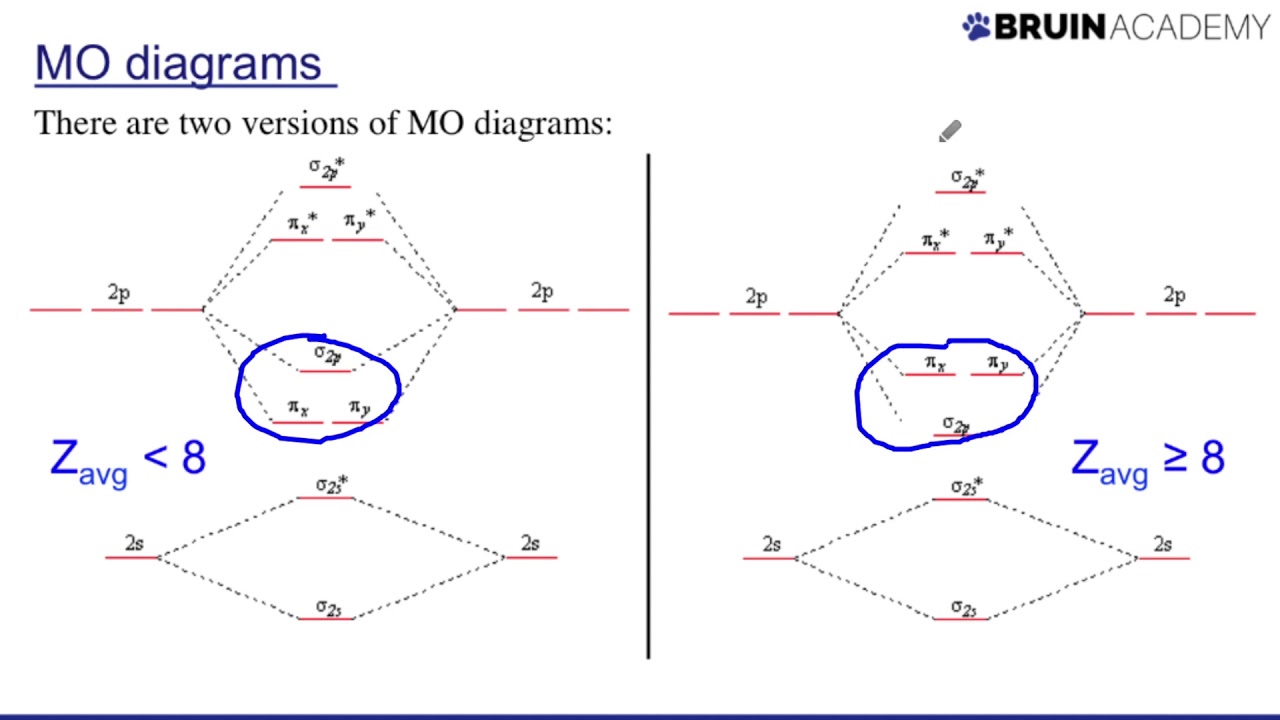
Drawing Molecular Orbital Diagrams - YouTube